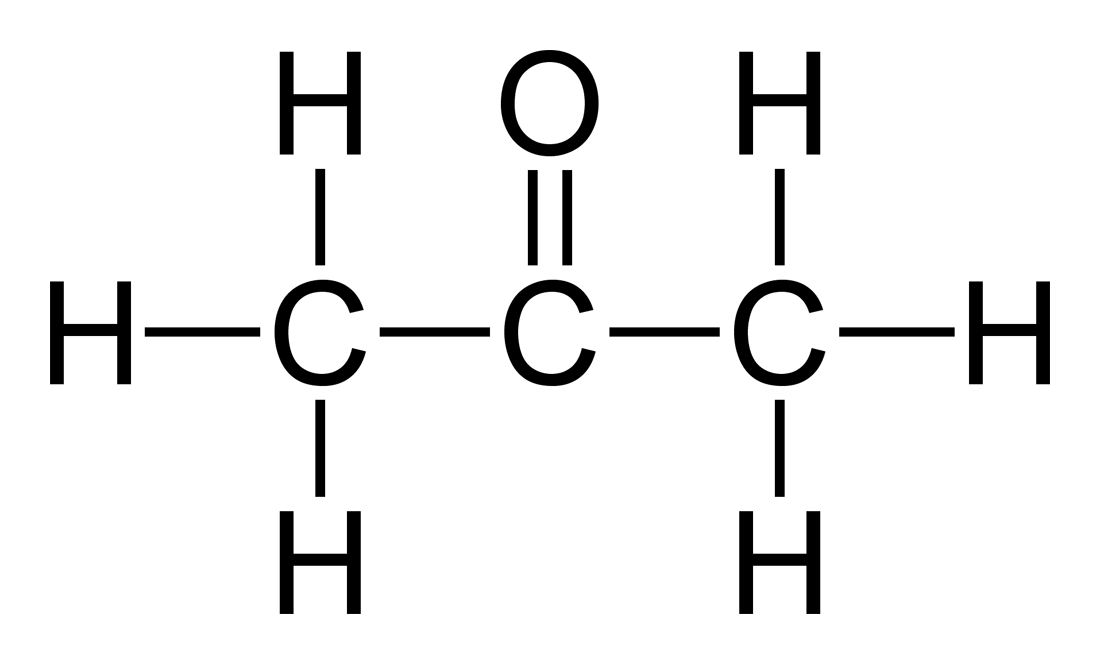
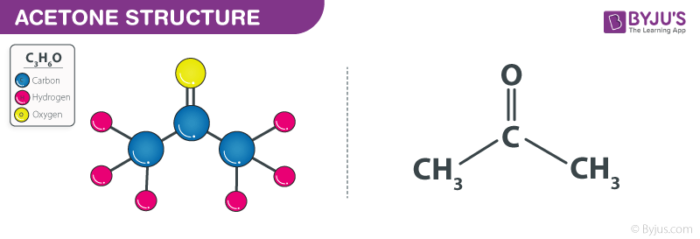
Solubility of nicotinic acid in water, ethanol, acetone, diethyl ether, acetonitrile, and dimethyl sulfoxide - ScienceDirect
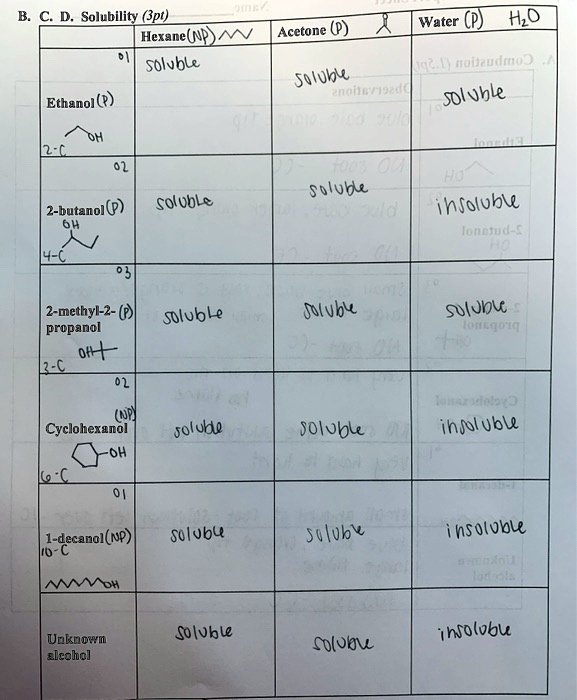
SOLVED: Text: Solubility (pt) Hexane (Not Soluble) Water (Soluble) Acetone ( Soluble) Ethanol (Soluble) 2-butanol (Soluble) 2-methyl-2-propanol (Soluble) Cyclohexanol (Soluble) 1-decanol (Not Soluble) Unknown alcohol (Soluble)

CLVIII.—The solubility of picric acid in mixed solvents. Part I. Water–alcohol and water–acetone mixtures - Journal of the Chemical Society (Resumed) (RSC Publishing)

Solubility of Paracetamol in Binary and Ternary Mixtures of Water + Acetone + Toluene | Journal of Chemical & Engineering Data
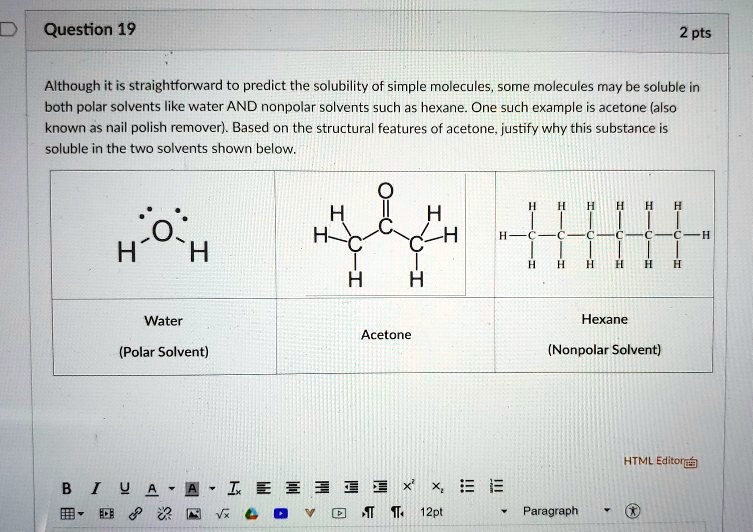
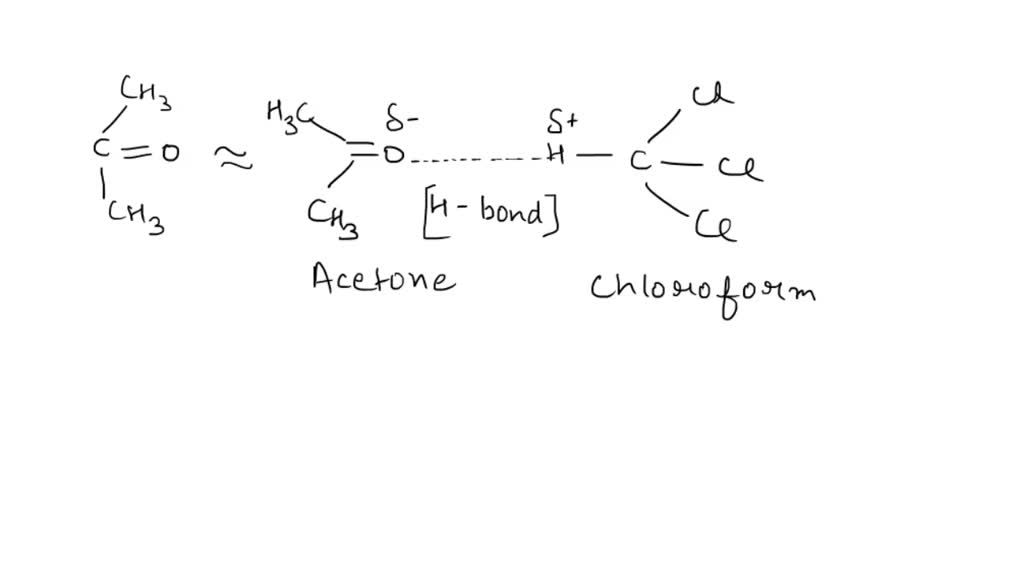
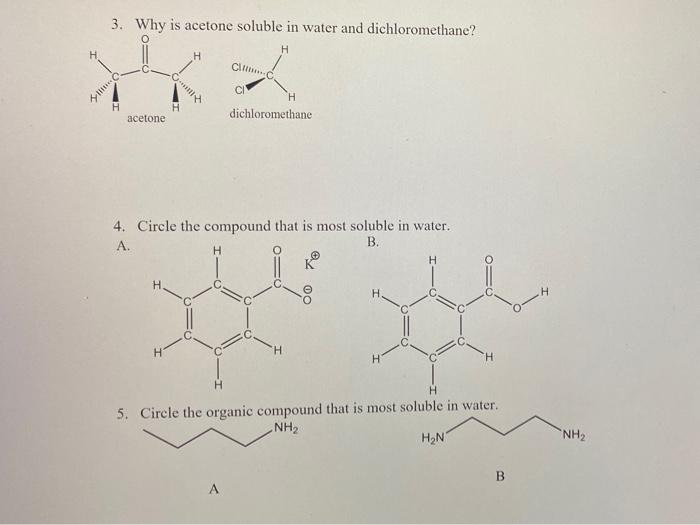
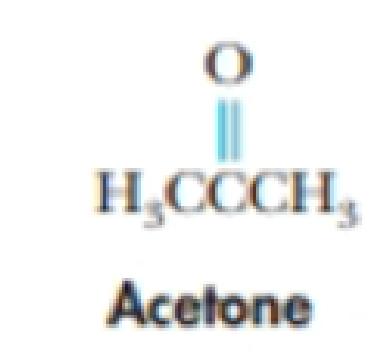
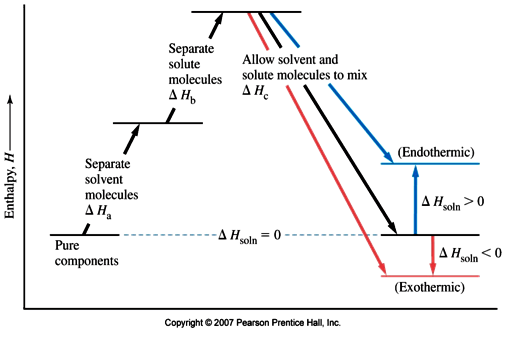
SOLVED: Although it is straightforward to predict the solubility of simple molecules, some molecules may be soluble in both polar solvents like water and nonpolar solvents such as hexane. One such example

Solubility of AZC as function of water/acetone mixture composition at... | Download Scientific Diagram

Acetone/Water Cosolvent Approach to Lignin Nanoparticles with Controllable Size and Their Applications for Pickering Emulsions | ACS Sustainable Chemistry & Engineering
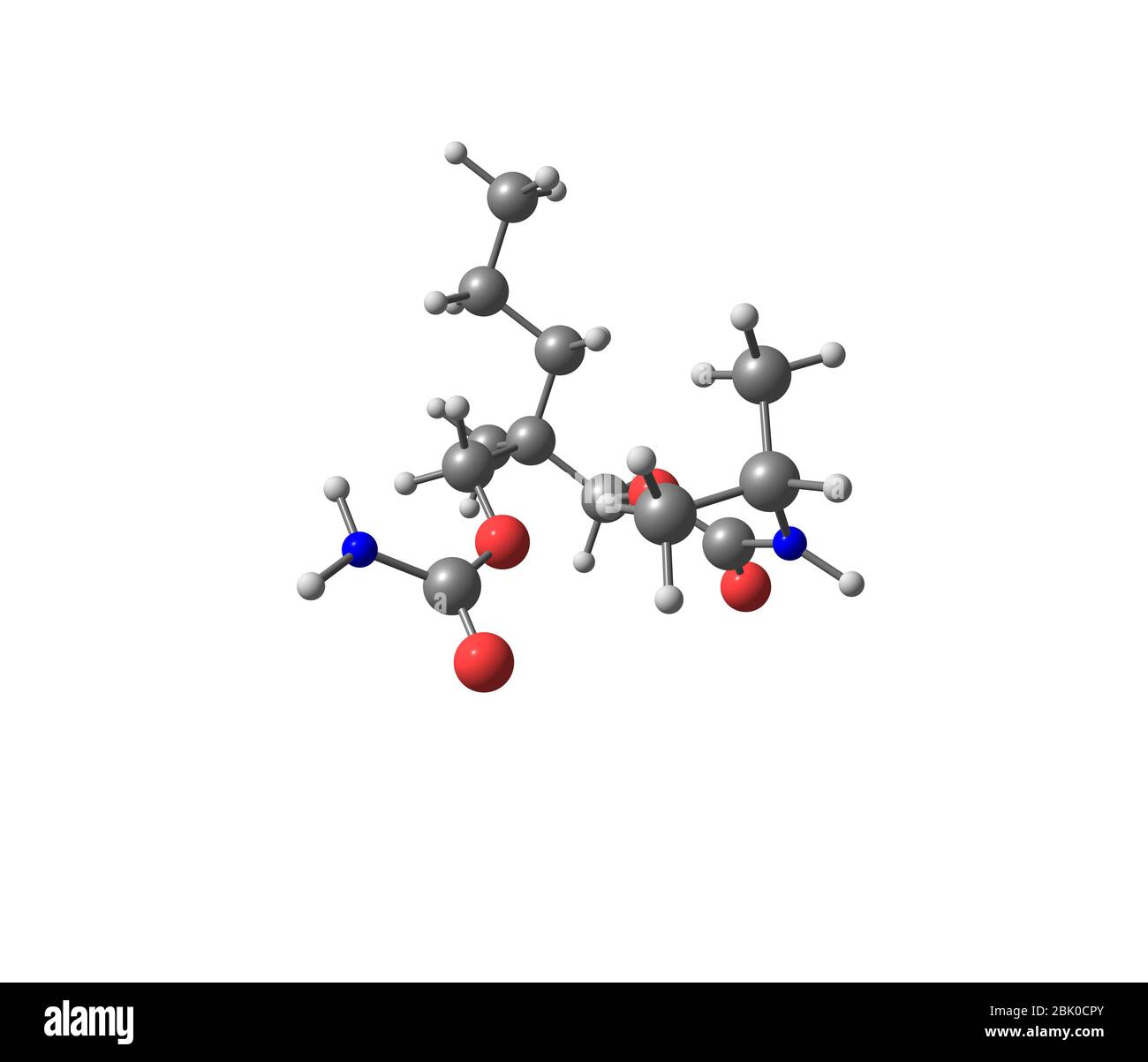
Carisoprodol is a centrally acting skeletal muscle relaxant. It is slightly soluble in water and freely soluble in alcohol, chloroform and acetone Stock Photo - Alamy

4) Me, SiCKOH) 72. Consider the following statements. 1. LiF is almost insoluble in water whereas LiCl is only soluble in acetone. II. Lil is soluble than Kl in ethanol III. BaO