![SOLVED: Acetone [(CH3)C=O] is a useful solvent because it dissolves a variety of compounds well. For example, both hexane [CH3(CH2)4CH3] and H2O are soluble in acetone. Explain why these solubility properties are SOLVED: Acetone [(CH3)C=O] is a useful solvent because it dissolves a variety of compounds well. For example, both hexane [CH3(CH2)4CH3] and H2O are soluble in acetone. Explain why these solubility properties are](https://cdn.numerade.com/ask_images/9d349811a4444b3a91bc31884ae75de9.jpg)
SOLVED: Acetone [(CH3)C=O] is a useful solvent because it dissolves a variety of compounds well. For example, both hexane [CH3(CH2)4CH3] and H2O are soluble in acetone. Explain why these solubility properties are

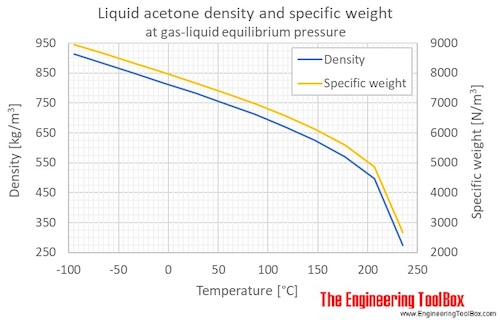
Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram

Shows the list of common organic solvents, their formula and melting... | Download Scientific Diagram

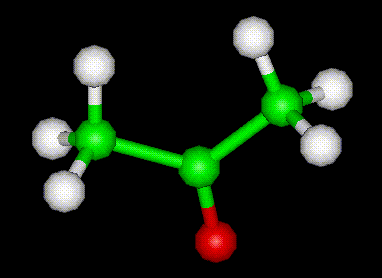
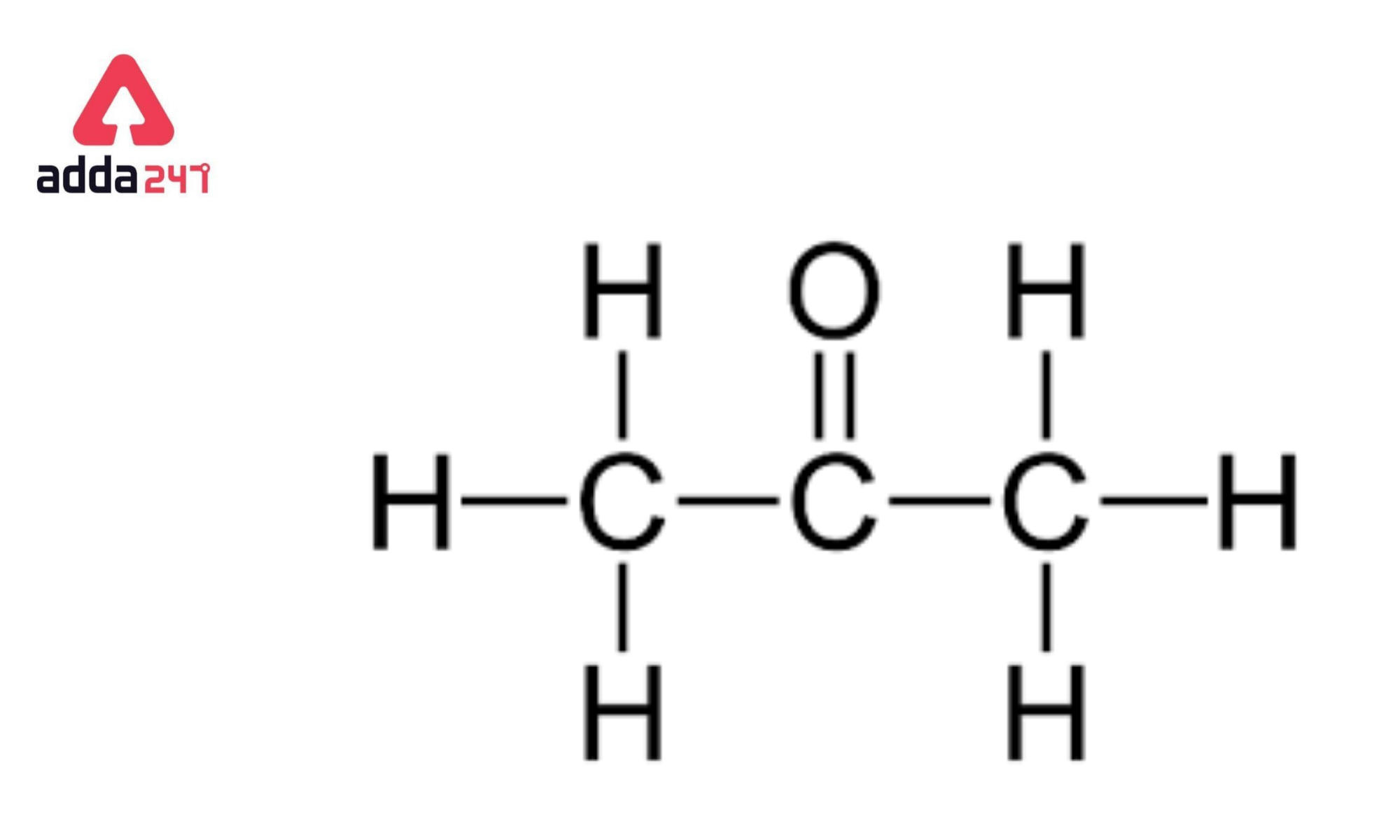
Acetone (CH3COCH3)- Structure, Properties, Preparation, Chemical properties, Uses and FAQs of Acetone (CH3COCH3)

















![Basic physical and chemical properties of acetone [9–11] | Download Table Basic physical and chemical properties of acetone [9–11] | Download Table](https://www.researchgate.net/publication/225596181/figure/tbl1/AS:393703542804517@1470877647990/Basic-physical-and-chemical-properties-of-acetone-9-11.png)